Melanoma : Sentinel Lymph Node Biopsy

Comments:
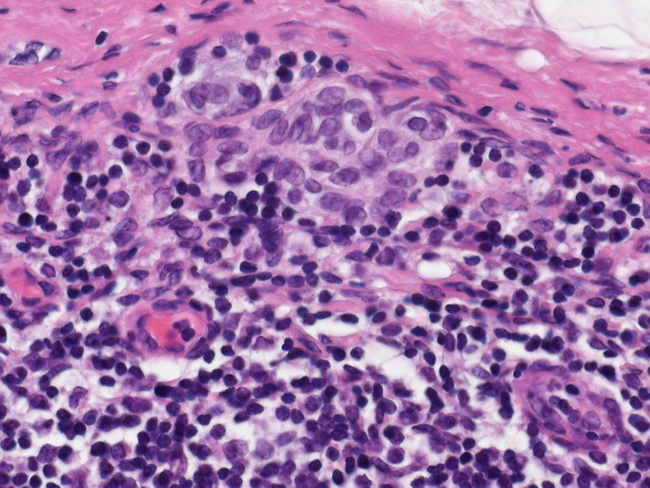
The removed sentinel lymph node is sent to pathology where it is bivalved or serially sectioned and processed for routine histology. If the routine H&E-stained step sections are negative, immunohistochemistry is performed with S-100 protein, HMB-45 (preferred), and/or MART-1 immunostains. The results of sentinel lymph node biopsy (SLNB) are incorporated into the AJCC staging system. PITFALLS: Benign nevus cell inclusions and histiocyte aggregates in lymph nodes can mimic metastatic melanoma. Capsular/subcapsular location, lack of cytologic atypia, and weak/absent staining for HMB-45 support nevus inclusions. Histiocytes lack nuclear atypia and are positive for CD68. CLINICAL UTILITY: SLNB status predicts the likelihood of tumor recurrence and patient survival in Stage 1 and Stage 2 melanoma. Patients with a negative SLNB and no clinical signs of other lymph node involvement can be spared regional lymph node dissection. The risk of regional lymph node recurrence in such patients is less than 4%. In patients with a positive SLNB, removal of the remaining regional lymph nodes is called Completion Lymph Node Dissection (CLND). A large multicenter randomized clinical trial (sponsored by NIH, NCI, John Wayne Cancer Institute) is underway to determine if CLND has therapeutic value for melanoma patients, specifically with regards to disease-free survival and melanoma-specific survival. The image shows melanoma micrometastasis under the capsule (near the top of the image). Image copyright: pathorama.ch.



